Abbott's FreeStyle® Libre 2 iCGM Cleared in U.S. for Adults and Children with Diabetes, Achieving Highest Level of Accuracy and Performance Standards - Jun 15, 2020
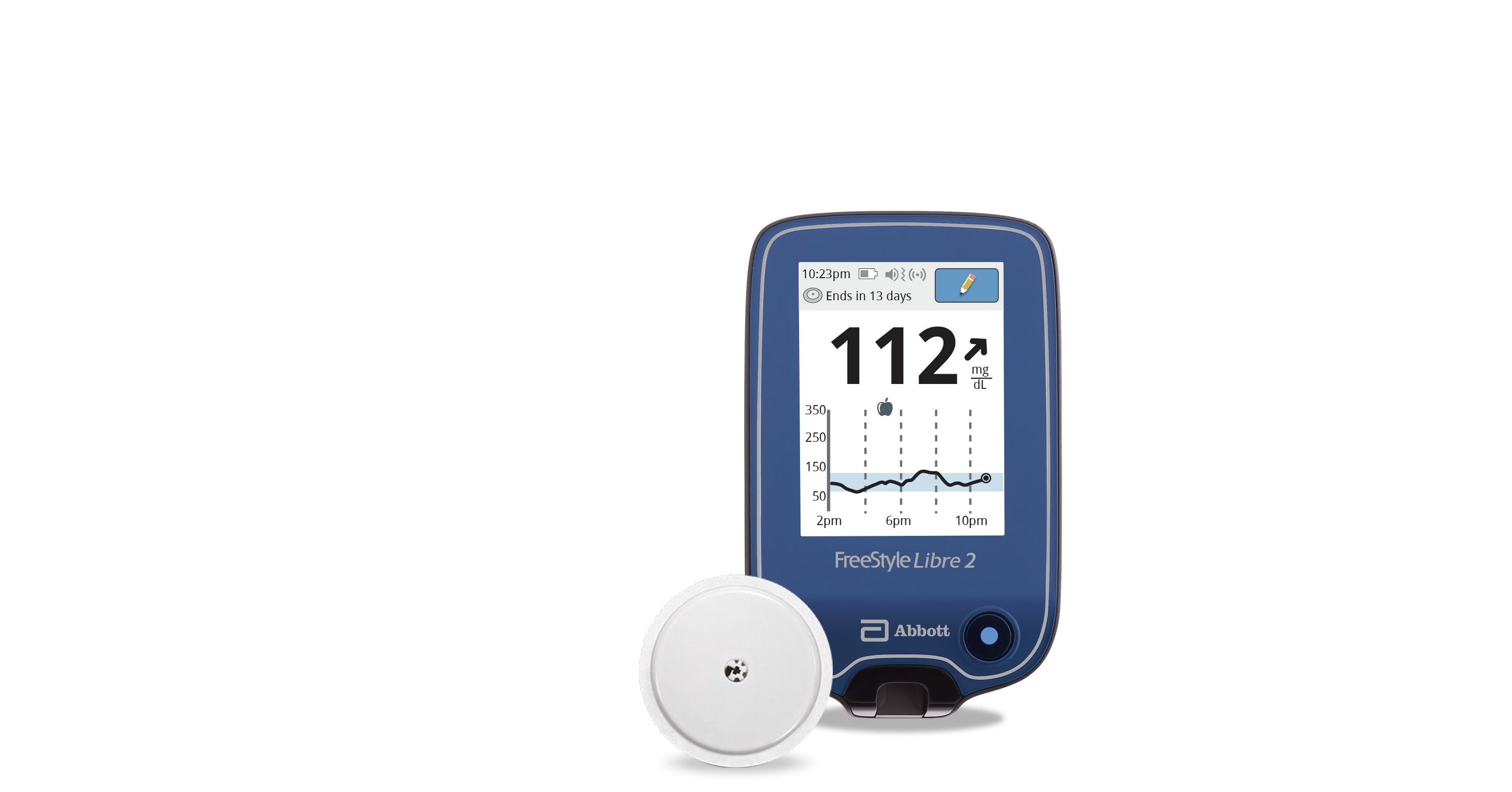
Abbott (NYSE: ABT), the worldwide leader in continuous glucose monitoring (CGM), announced today the U.S. Food and Drug Administration (FDA) cleared its next-generation FreeStyle® Libre 2

Diabetes Technology Meeting 2020 - Trisha Shang, Jennifer Y. Zhang, B. Wayne Bequette, Jennifer K. Raymond, Gerard Coté, Jennifer L. Sherr, Jessica Castle, John Pickup, Yarmela Pavlovic, Juan Espinoza, Laurel H. Messer

FDA Approves FreeStyle Libre 2 iCGM System for Patients With Diabetes
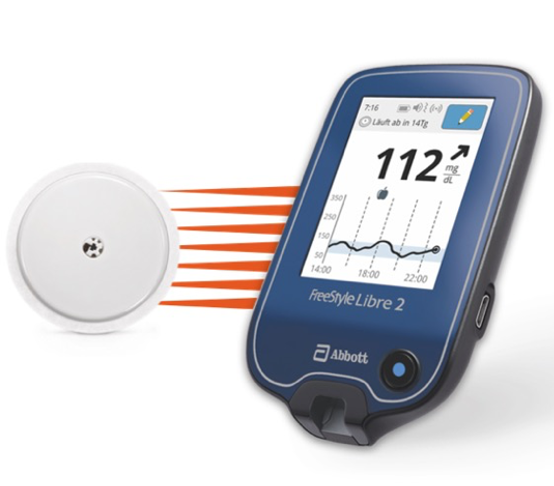
FreeStyle Libre 2 Cleared by FDA as iCGM
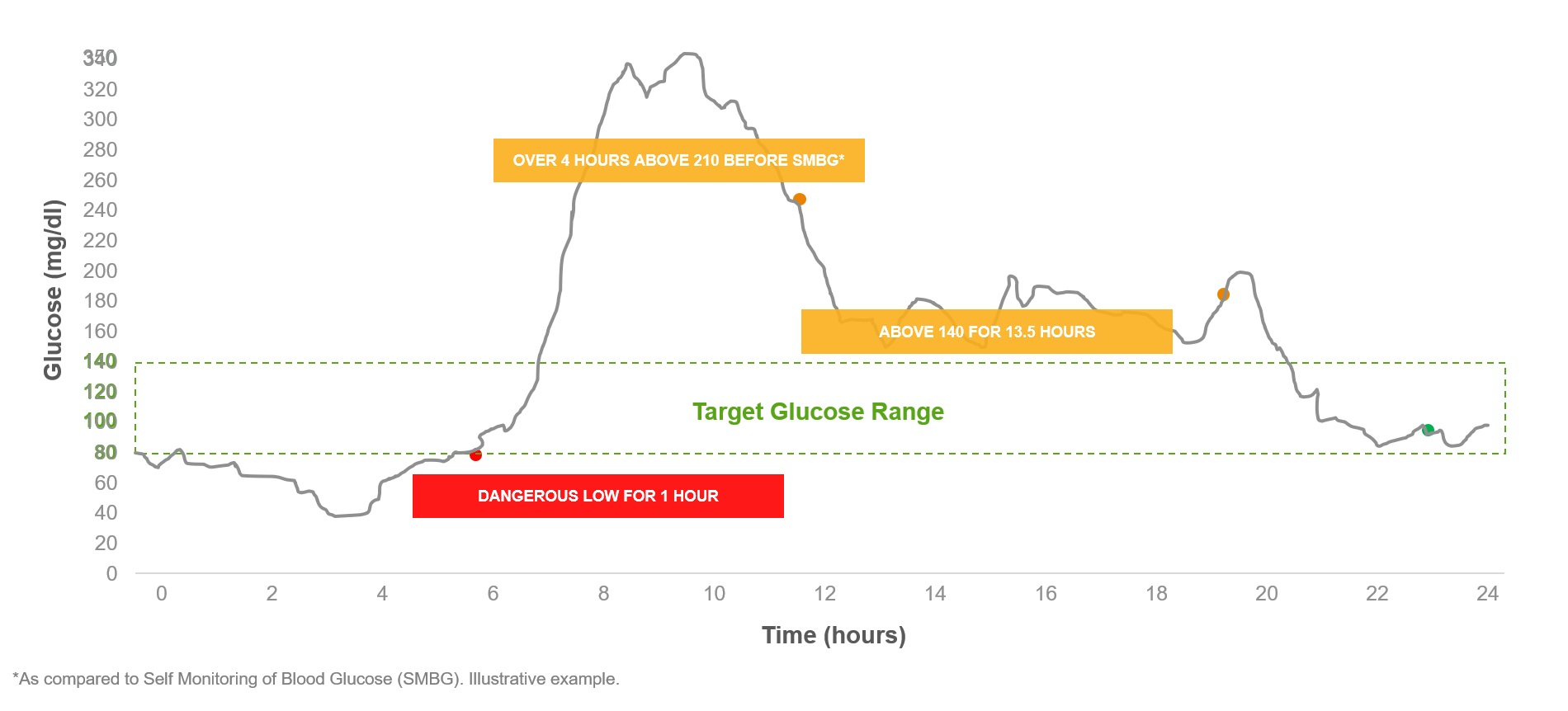
dxcm-20201231

Emerging Diabetes Technologies: Continuous Glucose Monitors/Artificial Pancreases
FreeStyle Libre 2 Continuous Glucose Monitor

FreeStyle Libre 2 System

Diabetes Technology Meeting 2020 - Trisha Shang, Jennifer Y. Zhang, B. Wayne Bequette, Jennifer K. Raymond, Gerard Coté, Jennifer L. Sherr, Jessica Castle, John Pickup, Yarmela Pavlovic, Juan Espinoza, Laurel H. Messer

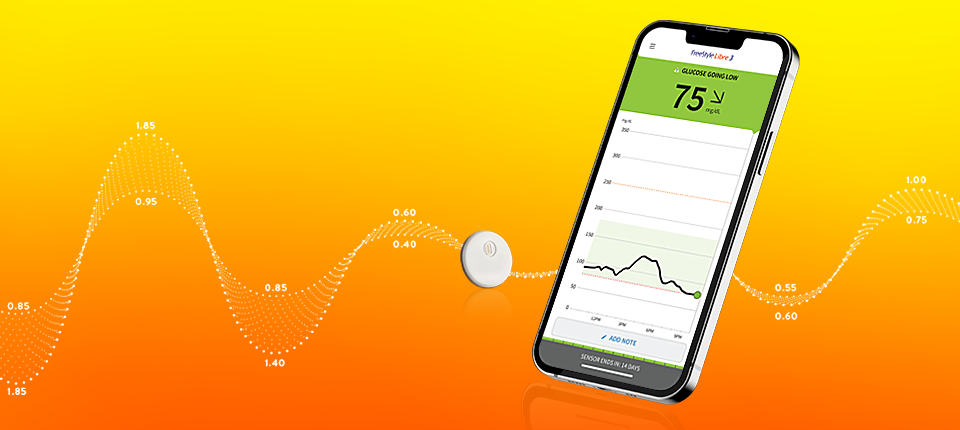
FreeStyle Libre 2 System (CGM)
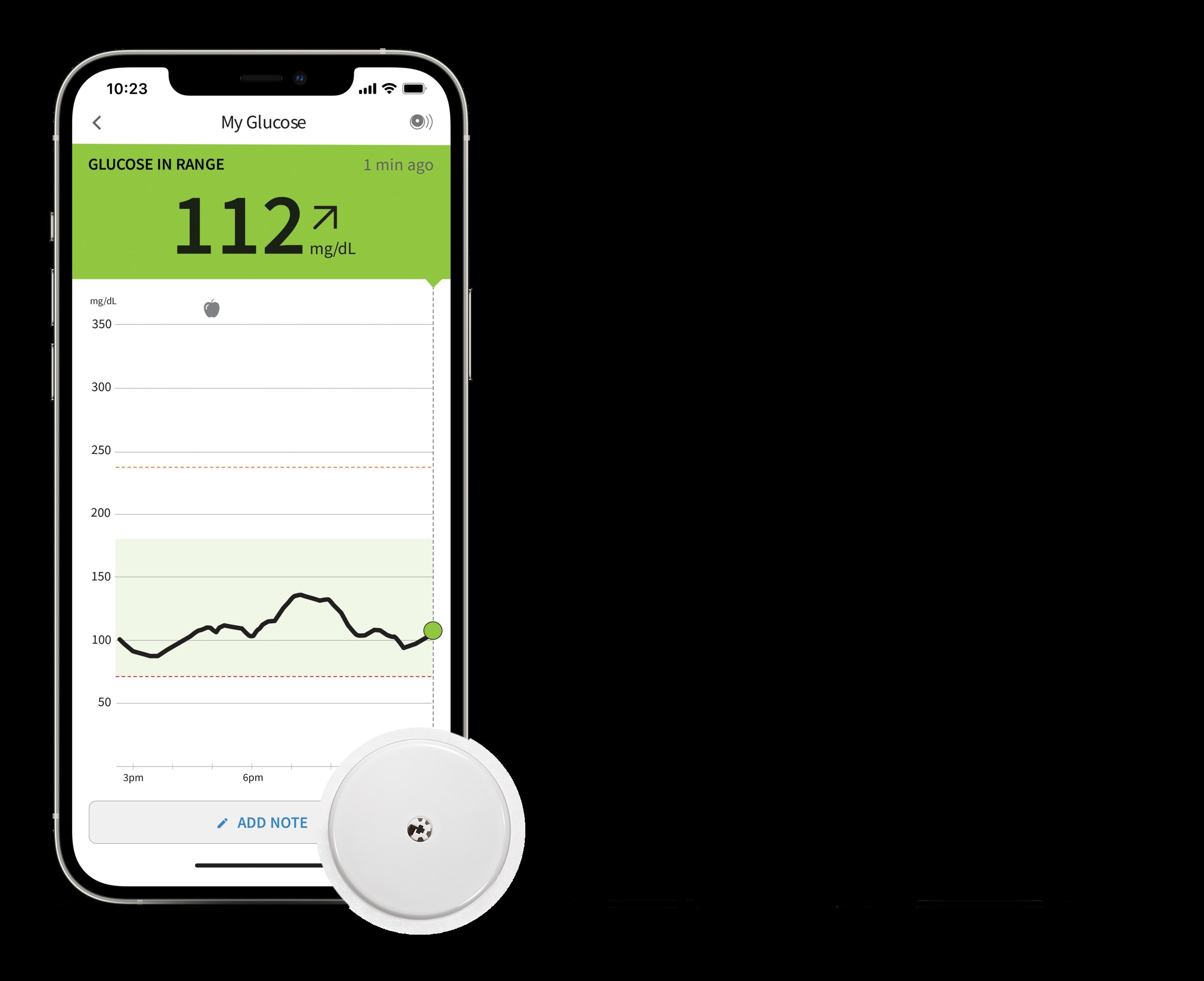
New Study Published in The New England Journal of Medicine Demonstrates Abbott's FreeStyle Libre® 2 System Has Positive Impacts on Glucose Levels and Quality of Life - Oct 6, 2022